Solubility of Calcium Carbonate in Ammonium Chloride Aqueous Solution at T = (298.15, 323.15, and 348.15) K | Journal of Chemical & Engineering Data

Uncovering the Role of Bicarbonate in Calcium Carbonate Formation at Near‐Neutral pH - Huang - 2021 - Angewandte Chemie International Edition - Wiley Online Library
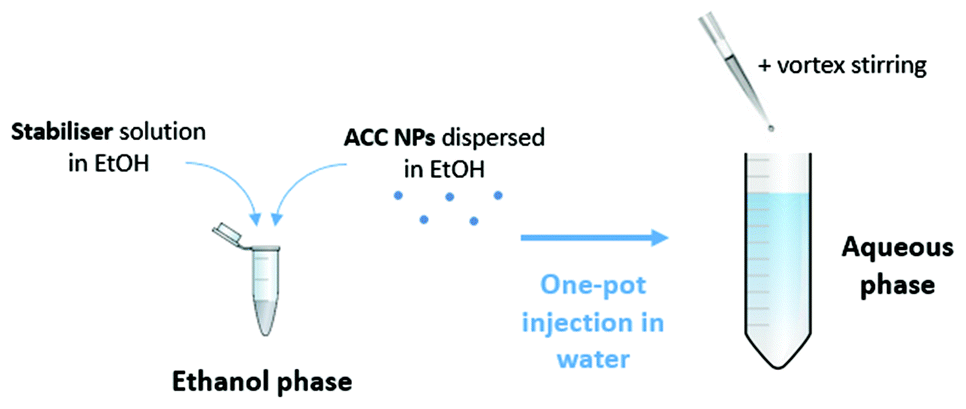
Eco-friendly processes for the synthesis of amorphous calcium carbonate nanoparticles in ethanol and their stabilisation in aqueous media - Green Chemistry (RSC Publishing) DOI:10.1039/D1GC03396D

Synthesis of calcium carbonate in a pure ethanol and aqueous ethanol solution as the solvent - ScienceDirect

Solubility of Calcium Carbonate in Ammonium Chloride Aqueous Solution at T = (298.15, 323.15, and 348.15) K | Journal of Chemical & Engineering Data

Pharmaceutics | Free Full-Text | Ground Calcium Carbonate as a Low Cost and Biosafety Excipient for Solubility and Dissolution Improvement of Praziquantel
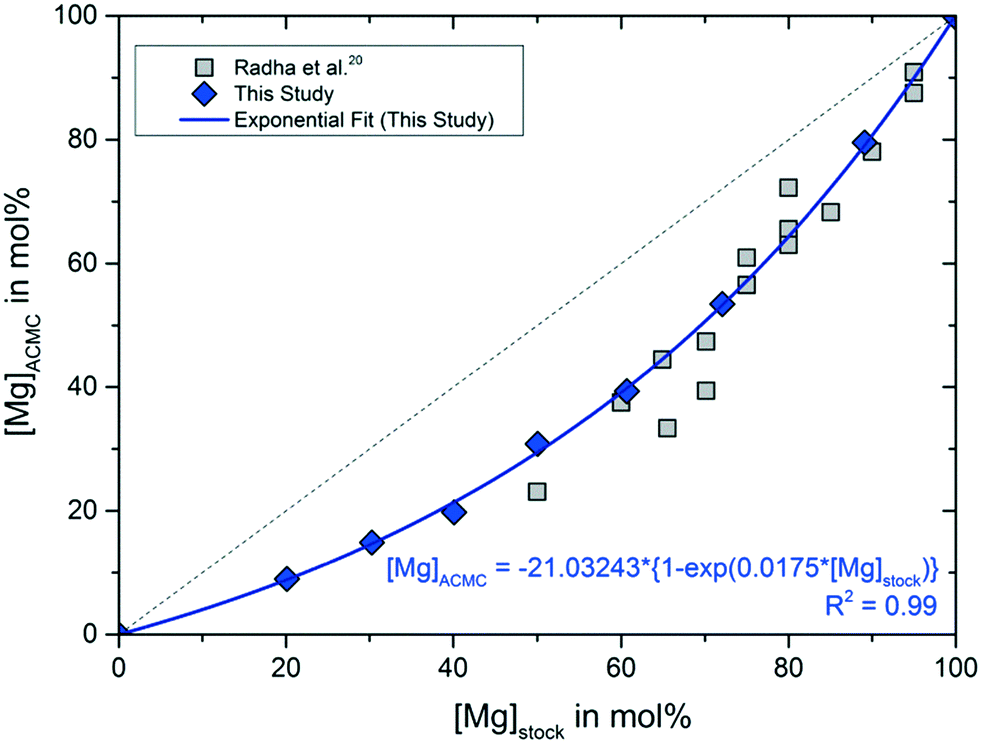
Solubility investigations in the amorphous calcium magnesium carbonate system - CrystEngComm (RSC Publishing) DOI:10.1039/C8CE01596A

Investigation of the stabilization of verdigris-containing rag paper by wet chemical treatments | Heritage Science | Full Text
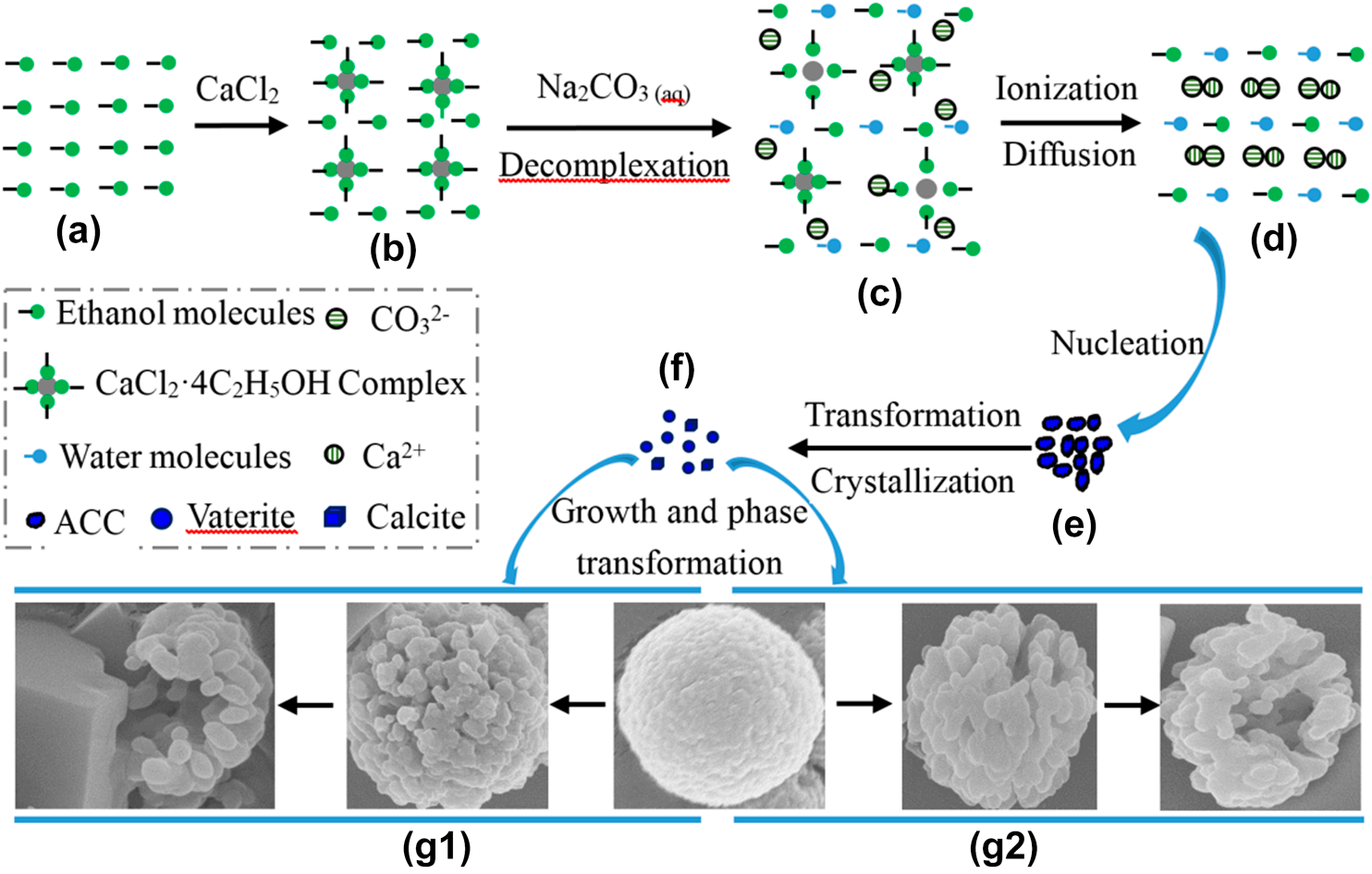
The advantage of alcohol–calcium method on the formation and the stability of vaterite against ethanol–water binary solvent method | Journal of Materials Research | Cambridge Core
Calcium Carbonate Particle Formation through Precipitation in a Stagnant Bubble and a Bubble Column Reactor

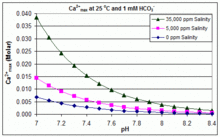
Solubility of calcium carbonate (lime scale) in water as a function of pH. | Download Scientific Diagram
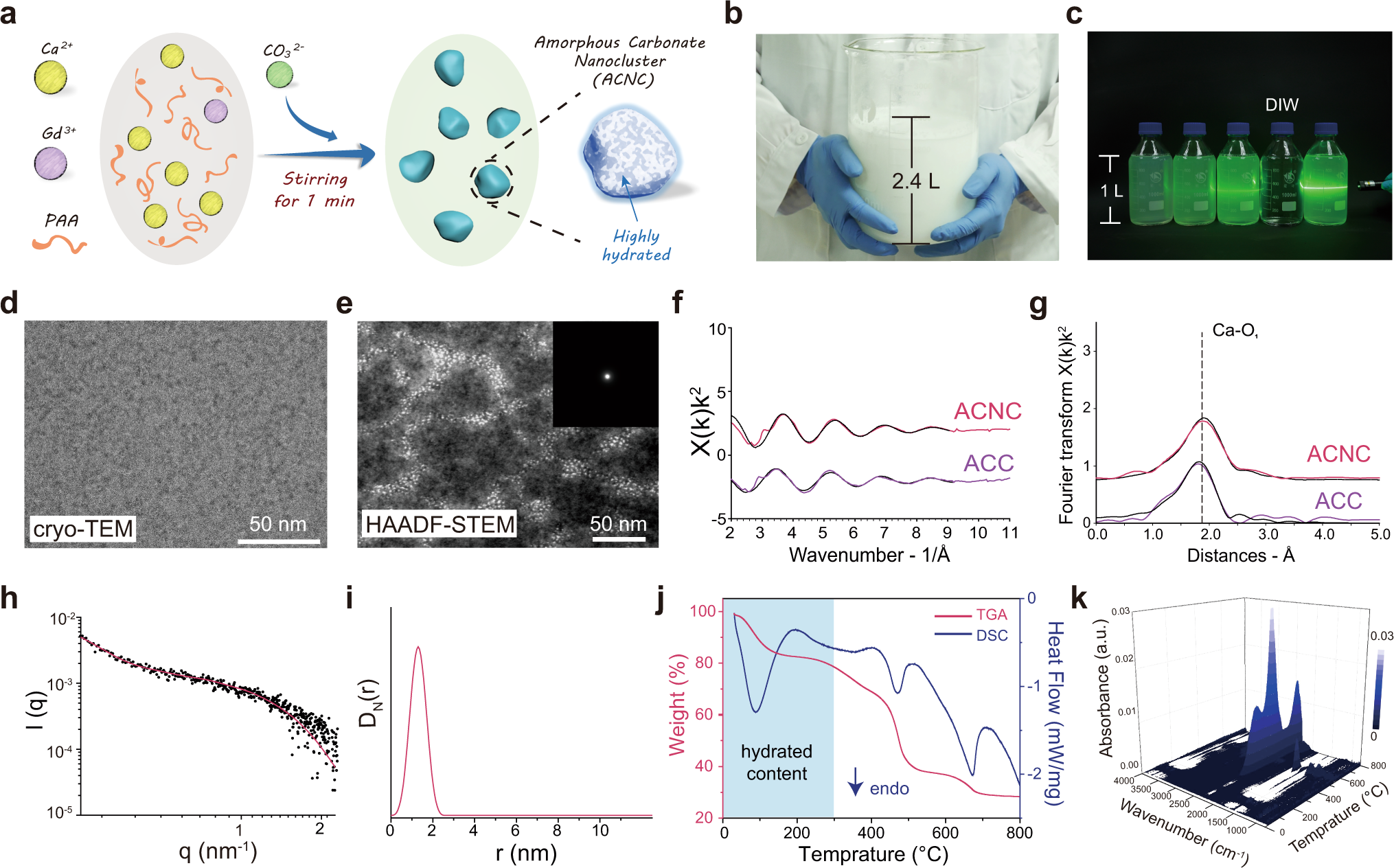
Highly hydrated paramagnetic amorphous calcium carbonate nanoclusters as an MRI contrast agent | Nature Communications
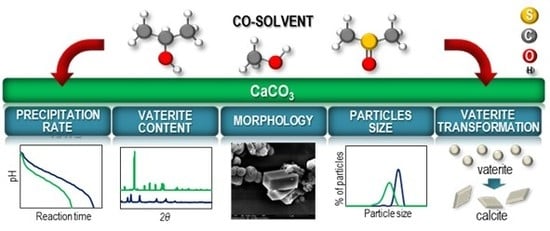
Materials | Free Full-Text | Precipitation and Transformation of Vaterite Calcium Carbonate in the Presence of Some Organic Solvents

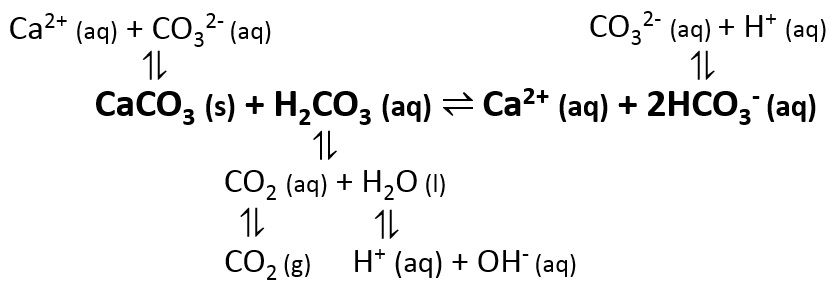

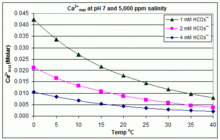
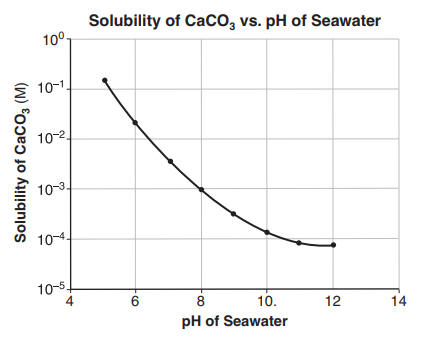
![Calcium carbonate solubility in function of pH [after (13)] | Download Scientific Diagram Calcium carbonate solubility in function of pH [after (13)] | Download Scientific Diagram](https://www.researchgate.net/publication/287707021/figure/fig8/AS:437295074549766@1481270678640/Calcium-carbonate-solubility-in-function-of-pH-after-13.png)