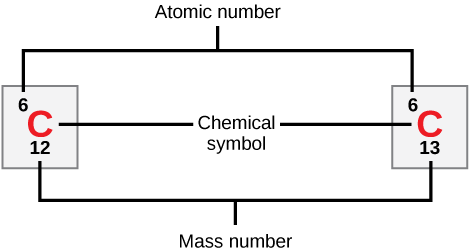
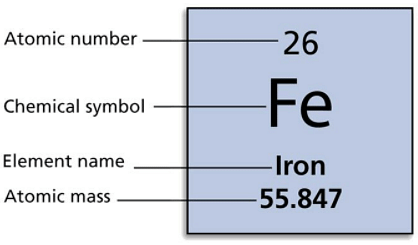
Calculating Particles for an ion. Representations from the Periodic Table Fe Iron Oxidation States Name Atomic Mass Atomic Number. - ppt download

Calculate the number of iron atoms in a piece of iron weighing `2.8 g` (Atomic mass of iron `= 56 u`
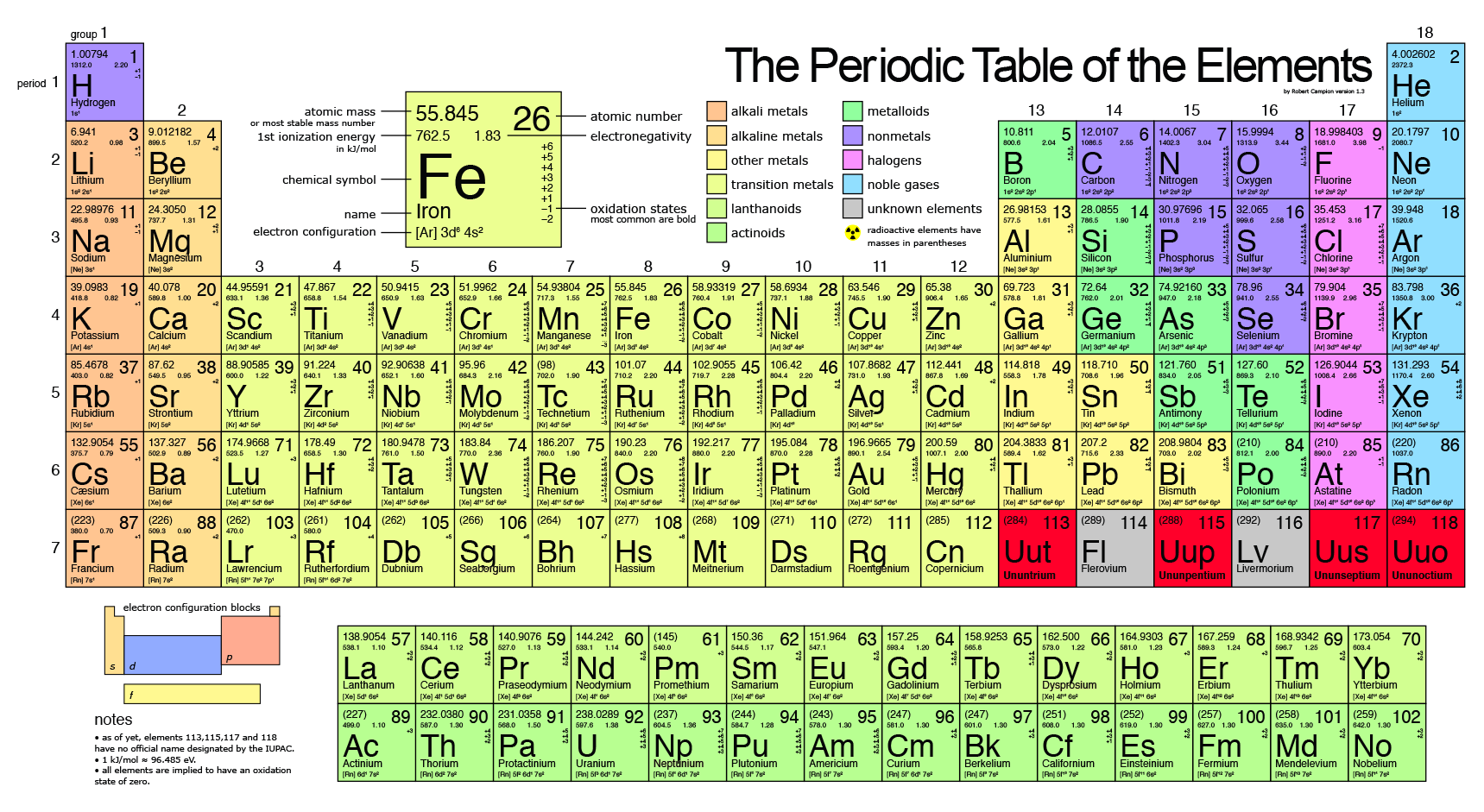
Why don't any of the isotopes of natural iron have the atomic mass of 55.85 amu, if the number listed in the periodic table is 55.85 amu? | Socratic

QUESTION: Which of the following has the largest mass number? A. 56Mn, B. 56Fe, C. 56Cr, D. they all have the same mass number The mass number is. - ppt download
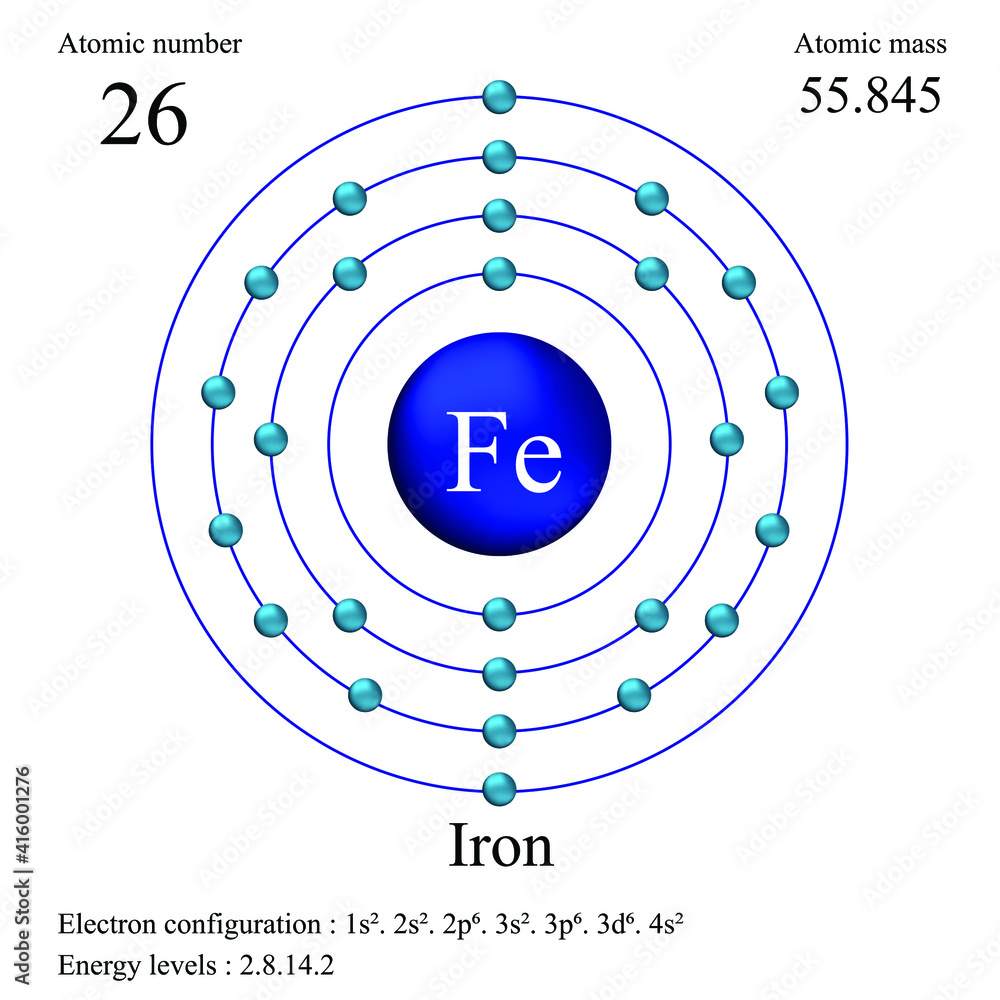
Iron atomic structure has atomic number, atomic mass, electron configuration and energy levels. Stock Vector | Adobe Stock

The atomic number and the mass number of an element is 26 and 56 respectively. Calculate the number of electrons, protons and neutrons in its atom. Draw the structure.