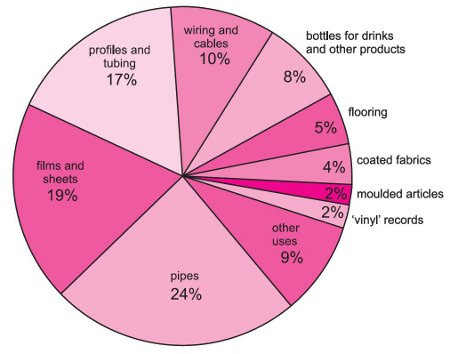
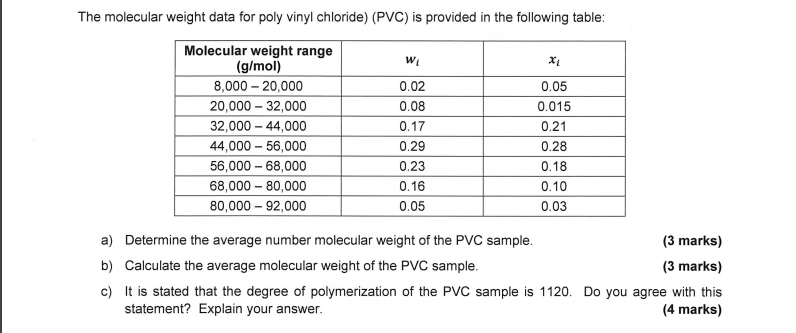
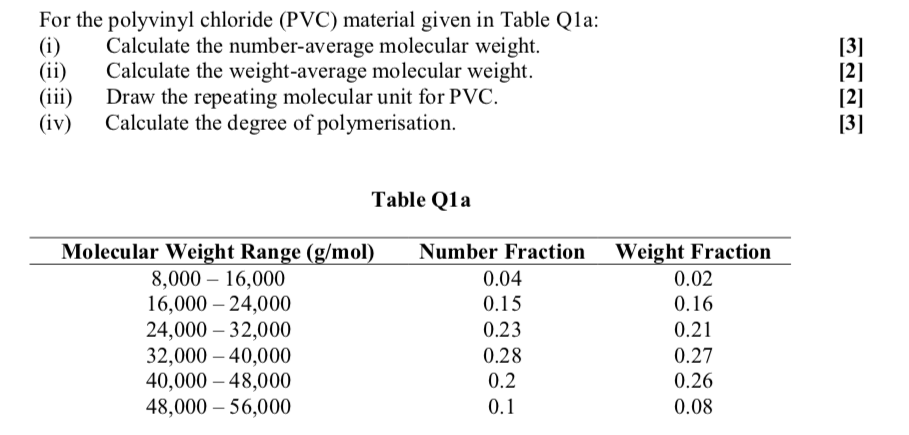
A solution containing 4.0 g of PVC in 2 litre of dioxane (industrial solvent ) was found to have an osmotic pressure 3.0xx10^(-4) atm at 27^(@)C The molecular mass of the polymer will be :

SOLVED: Solutions of polyvinyl chloride (PVC) in cyclohexanone at 298 K were found t0 have the following solution heights for the given mass concentrations 9 L– hcm -00 0.30 2.00 0.75 3.00

Comparison of glass transition temperature T g vs molecular mass M:... | Download Scientific Diagram
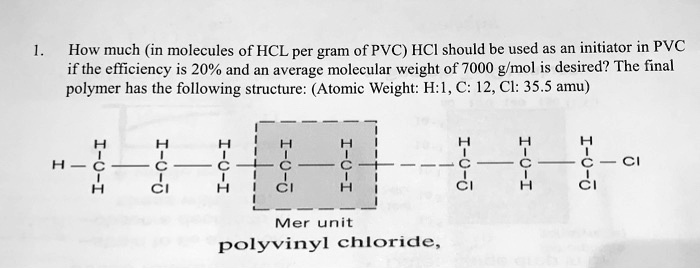
SOLVED: How much (in molecules of HCL per gram of PVC) HCI should be used as an initiator in PVC if the efficiency is 20% and an average molecular weight of 7000
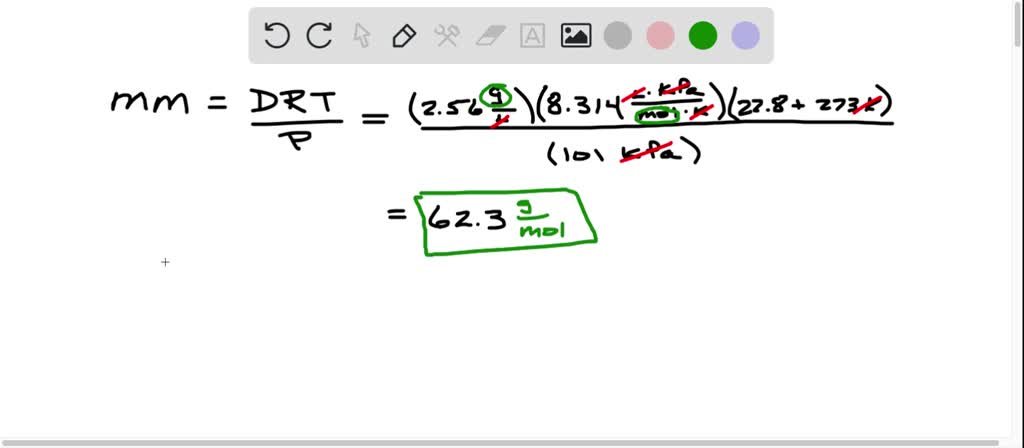
SOLVED: Monochloroethylene gas is used to make polyvinylchloride (PVC). It has a density of 2.56 g / L at 22.8^∘C and 101 kPa. What is the molar mass of monochloroethylene? What is

Chemical modification of PVC by different nucleophiles in solvent/non-solvent system at high temperature | SpringerLink
![The osmotic pressure of a solution containing 40 g of solute (molecular mass 246) per litre at 27^oC is: [R = 0.0822 atm L mol^-1] The osmotic pressure of a solution containing 40 g of solute (molecular mass 246) per litre at 27^oC is: [R = 0.0822 atm L mol^-1]](https://dwes9vv9u0550.cloudfront.net/images/1584107/8a075a92-5b07-4da2-98a8-7e2ad8c48a64.jpg)
The osmotic pressure of a solution containing 40 g of solute (molecular mass 246) per litre at 27^oC is: [R = 0.0822 atm L mol^-1]

All-PVC” Flexible Poly(vinyl Chloride): Nonmigratory Star-Poly(vinyl Chloride) as Plasticizers for PVC by RAFT Polymerization | Macromolecules
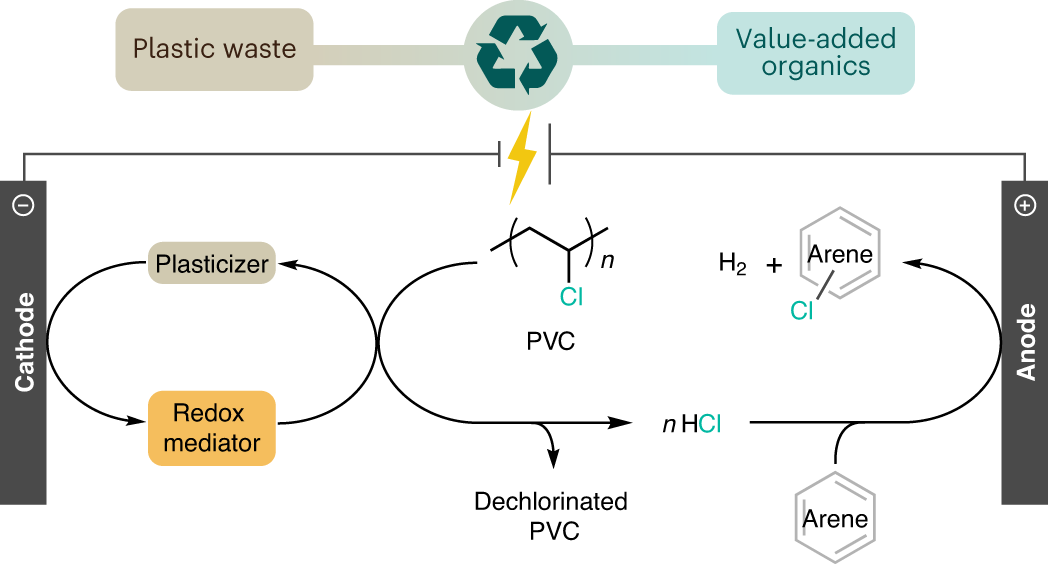
Using waste poly(vinyl chloride) to synthesize chloroarenes by plasticizer-mediated electro(de)chlorination | Nature Chemistry